The instructor could create a video performing the demonstration and share the video with the students.The instructor will perform the demonstration in-person and the students will record their observations and conclusions on the handout.Physical and Chemical Changes Demonstration: The formation of a precipitate (a solid is produced out of solution).A chemical change is also called a chemical reaction. A chemical change is a change in the composition of the original substance. In the simplest sense, a physical change is a change in the form of the original substance. In chemistry, the term change can refer to both physical and chemical changes. The video could be then be shared with the students in the LMS or some other online platform. Another option is for the instructor to record a video performing the demonstration. The demonstration can be performed in person and the students can complete the demo handout during the demonstration. It is a good idea to record the indicators of chemical changes on the board as you review the background information with the class. Review the background information with students before completing the demonstration. Scientific Practices: Constructing ExplanationsĮssential Question : How can you distinguish between a physical and a chemical change? Whereas, the part of the wax close to the flame actually burns and gives light and carbon dioxide, which is irreversible and a chemical : Chemical Reactions - The face that atoms are conserved, together with the knowledge of the chemical properties of the elements involved, can be used to describe and predict chemical reactions. During the burning of a candle, the melting of wax is a physical change. There is an example where physical and chemical change takes place simultaneously. So, both physical and chemical properties get changed, during a chemical change as a completely new product is formed which has no relation with the original reactant. Although chemical changes could also be recognized by a sign like odor, color change, or production of a gas, all of those indicators may end up from phase transition. Many chemical changes are irreversible, and lots of physical changes are reversible, but reversibility can not be classified as a criteria to differentiate. Many physical changes also involve the rearrangement of atoms which are mostly noticed during crystal formation. A steel blank is repeatedly heated and hammered which changes the hardness of the steel, its flexibility and its ability to take care of a pointy edge, so on application of force and energy, the form is modified An example of a phase transition is that the process of forming a blade by tempering steel. 
For instance, salt dissolved in water is often reversed by allowing the water to evaporate.Īn activity is absolute to involve a change within the physical properties. Generally, a natural action is reversible using physical means, like mechanical force or pressure. This contrasts with the concept of the chemical process within which the composition of a substance changes or one or more substances combine or cut to create new substances. 
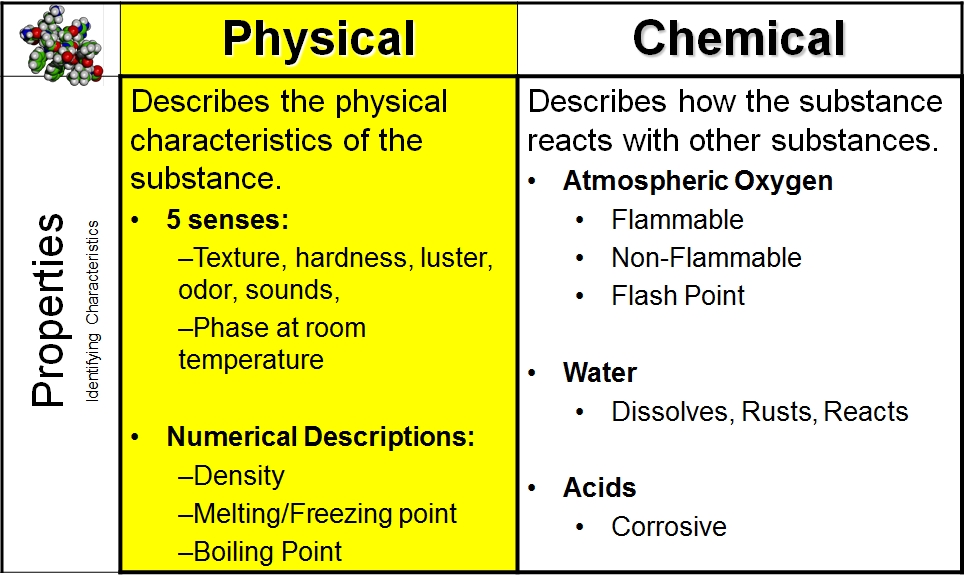
Physical changes occur when objects or substances undergo a sort of change that doesn't change their composition chemically. Physical changes are wont to separate mixtures into their component compounds, but cannot usually be wont to get into the molecular/atomic level and separate compounds into chemical elements. Physical changes are changes that affect the shape of a chemical substance, but don't affect its chemical composition. Let us study physical and chemical changes. In a chemical change, properties of the substance change, and also the properties of the matter. Hint: When a chemical change occurs, a completely new product is formed, and the process cant be reversed back too. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
